Catalysts
PRODUCTS
-
PHCbi brand’s LabSVIFT® IoT lab management solution is designed to provide real-time visibility and control of laboratory equipment and environments across single or multiple facilities. This cloud-based platform enables centralized monitoring, customizable alerts, and secure data access via a web-based dashboard. The new health check* function continuously evaluates equipment performance, identifies irregularities, and provides recommended corrective actions to maintain optimal operation. The system supports FDA 21 CFR Part 11 compliance and is compatible with select PHCbi and third-party devices. *health check function compatibility varies by model
-
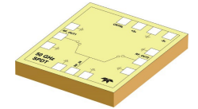
CPI EDB offers the 3469 magnetron transmitter system for use as a high-power source of RF power for outdoor-mounted radar applications. The transmitter cabinet contains the high voltage power supply, solid-state switch, and the high voltage tank assembly which includes the pulse transformer, energy storage high voltage capacitor, and filament power supply.
-
Teledyne HiRel Semiconductors has announced the release of the TDSW050A2T, a high-performance, rad-tolerant RF switch designed for demanding aerospace and defense applications.
-
Meaningful insights. Better results. Smarter utility management. Trimble Unity software platform offers a suite of applications and tools to support smart water management. Includes advanced workflows to map, manage, measure and improve asset performance.
-
Learn how robust in-house analytical services and protein characterization are crucial for successful development and manufacturing programs.
WHITE PAPERS AND CASE STUDIES
-
Transferring From Microfluidics To A Turbulent Mixer
What solutions did the client featured in this case study find when they sought help in transferring their formulation process to a turbulent Impinged Jet Mixing (IJM)?
-
RO Protection At Formosa Plastic Group, Taiwan
In this case study, learn why Formosa Plastics Group in Taiwan used Atlantium Technologies' HOD UV system to protect RO membranes from biofouling, resulting in cost savings and plans for further installations.
-
Why A Leading Manufacturing Partner Chose NeoTOP Packaging Machines
Learn how a leading CDMO partnered with Körber to quickly deploy a high-speed packaging solution to meet tight production deadlines in the face of a global pandemic.
-
Implementing A Next-Gen MES At A Fraction Of The Cost
Discover how a top medical device manufacturer accelerated product launch by digitizing assembly processes, streamlining training, and ensuring compliance.
-
Disinfection For Drinking Water Plant In Chennai, India
The Chennai Metropolitan Water Supply and Sewerage Board (CMWSSB) encountered significant challenges in treating high algae loads at its drinking water plant, which processes 1.5 million liters of water daily. The primary issue arose from the organic load in lake water, which, when treated with conventional chlorine disinfectants, resulted in harmful disinfection byproducts (DBPs).
-
Strategic Pre-IND Consulting For Gene Therapy In Rare Leukemia
Learn about the collaborative efforts of an innovative biotech startup and Ergomed Consulting to overcome challenges in the pre-IND phase for a novel gene therapy targeting a rare form of leukemia.
-
Transforming PSP Enrollment To Enhance User Experiences And Optimize Workflows
Lengthy enrollment calls burden patients and PSP teams alike. See how a digital-first intake approach cut onboarding time and improved outcomes for everyone involved.
-
Accelerating Early Phase Oncology Study Development
Explore a close collaboration to co-develop a comprehensive, adaptive protocol designed to support key decisions across multiple trial phases for an innovative cancer therapy.
-
Flexible Integrations To Reduce Risks And Increase Efficiencies
Learn how a trial team kept the blind of central lab samples and patient treatment across a Phase III and extension trial, whilst optimizing the supply chain.
-
Quick And Easy Pipe Repair In Virginia Saves Thousands Of Dollars
Learn how JCSA efficiently replaced ten dresser-style couplings on a 16" steel pipe spanning a 600-foot bridge using the versatile HYMAX VERSA coupling, enabling a quick and cost-effective repair without removing the pipe.
-
ArxZyme™ Acetone Removal For AMPAC Fine Chemicals
AMPAC faced a critical challenge with acetone management. The high acetone levels in the effluent from its chemical processing operations required an effective industrial wastewater treatment solution to meet environmental regulations and ensure safe discharge.
-
How Fine Screens Simplify The New Waste Discharge Requirements
How are fine screen solutions aiding winemakers in their efforts to meet California State Water Resources Control Board mandates for enhanced monitoring and treatment of winery process water.
NEWS
-
Consulta Remedios Selects Kevel's Retail Media Cloud To Power Its Next-Generation Advertising Platform5/2/2025
Kevel, the leading provider of API-based retail media ad serving technology, today announced that Consulta Remédios, Brazil's largest medicine price comparison platform, has selected Kevel's Retail Media Cloud to power the development and enhancement of its in-house retail media network.
-
Drawing Inspiration From Ancient Chemical Reactions3/20/2025
To help find solutions to the planet’s climate crisis, MIT Associate Professor Daniel Suess is looking to Earth’s ancient past.
-
Be Part of Photonics Future: CIOE 2025 Visitor Registration Live Now!4/9/2025
The 26th China International Optoelectronic Exposition (CIOE 2025) is set to take place from September 10–12, 2025, at the Shenzhen World Exhibition & Convention Center, spanning 240,000 square meters of exhibition space.
-
Pocket Alarm Detects And Records RF/Microwave Directed Energy Weapons, Nuclear Events, And Terrestrial Gamma Bursts5/8/2025
Ultraview Corp’s EM/GAMMALERT is a new pocket device that warns of RF/microwave jammer and directed energy weapon deployment1 - an emerging threat speculated as a modality in “Havana Syndrome”.
-
3-In-1 Orbital Thread Milling Cutters For New Thread Sizes1/14/2025
Following the successful introduction of the Thrill·tec TC645 Supreme orbital drill/thread mills for dimensions from M4 to M12 in 2023, Walter is now expanding its range to cover additional metric dimensions from M14 to M20 as well as their imperial equivalents.
ABOUT
Catalysts
Catalysts work by changing the structure of a molecule or by bonding to reactant molecules causing them to combine, react, and release a product or energy. For example, a catalyst is required in order for oxygen and hydrogen to bond, combine and produce water.
Catalysts increase the rate of chemical reactions. Unlike a reagent that gets consumed as apart of the chemical reaction, catalysts do not get consumed in the reaction. They can actually participate in multiple chemical transformations.Catalysts can be organic, synthetic or metal.
All processes need energy to take place. Processes require a very high level of energy in order to spark or start if a catalyst isn’t present. When a catalyst is present the amount of energy required to spark the reaction is lowered and that makes the reaction happen faster and more efficiently.
Catalytic effects vary depending on other substances present that are a part of the chemical reactions. There are certain substances that can inhibit the effectiveness of a catalyst like poisons, and other substances that promote or increase the effect of the catalyst. Inhibitors or negative catalysts are very important in medicine to treat mental illnesses, high blood pressure, cancer and many other diseases.
Catalysts are often used to help crack, or split, larger organic molecules like hydrocarbons into simpler molecules. It does this by breaking carbon-carbon bonds. The speed of the splitting process depends heavily on what specific catalyst is used and the temperature. Applications of this technology include oil and petrochemical industries.